RTA-102
Catalogue number: RTA-102
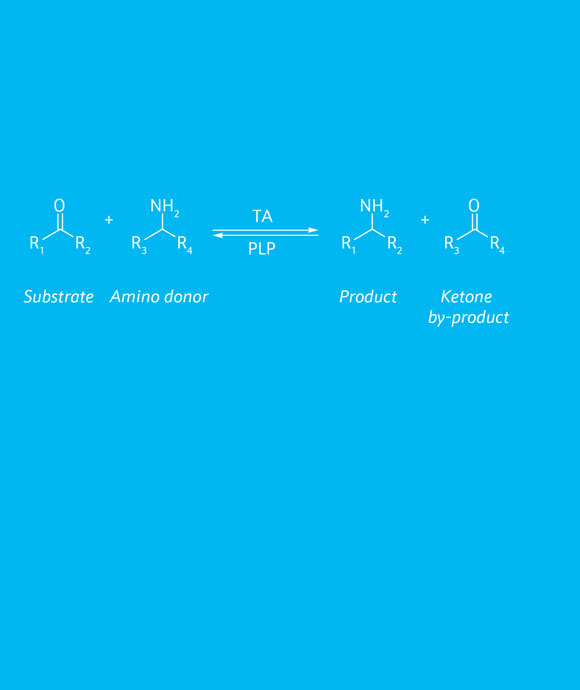
Aromatic and aliphatic primary amines can be obtained using our transaminases (TAs) enzymes for the formal reductive amination of ketone to the corresponding (R)- or (S)-amine. The results will depend on the inherent enzyme substrate scope but also on the substrate properties. In fact, if the amine moiety in the product molecule is stabilised by hydrogen bonds with other neighbouring functionalities, the reaction will reach higher conversions because the concurrent deamination reaction is less favoured.
TAs require a PLP cofactor for catalysing the reaction. PLP binds in the active site of the transaminase and it is instrumental for catalysing the transfer of the amine from an amino donor to the substrate, hence forming the desired chiral amine.